Blog
Mr. Yuk™’s Influence on Child-Resistant Packaging
Read Blog


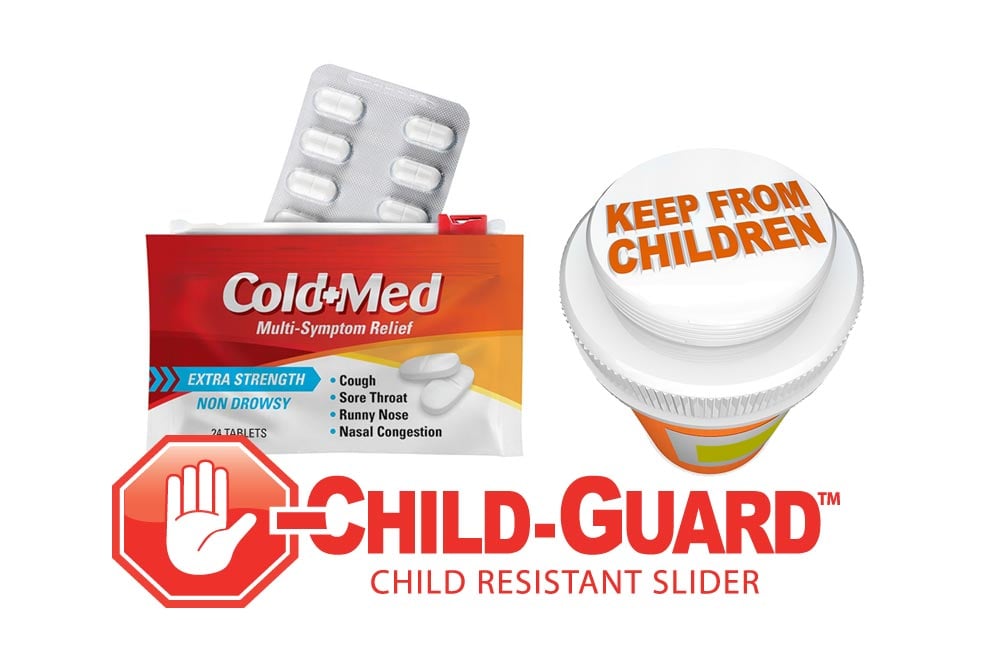
Child-resistant packaging and closures are vital to protecting our children. Our new, innovative Child-Guard® slider and child-resistant zipper are specially designed for products that should not be handled by children without adult supervision. Now, you can enjoy the benefits of flexible packaging with proven child-resistant reclosability while meeting the convenience expectations of consumers.
Explore Products





